 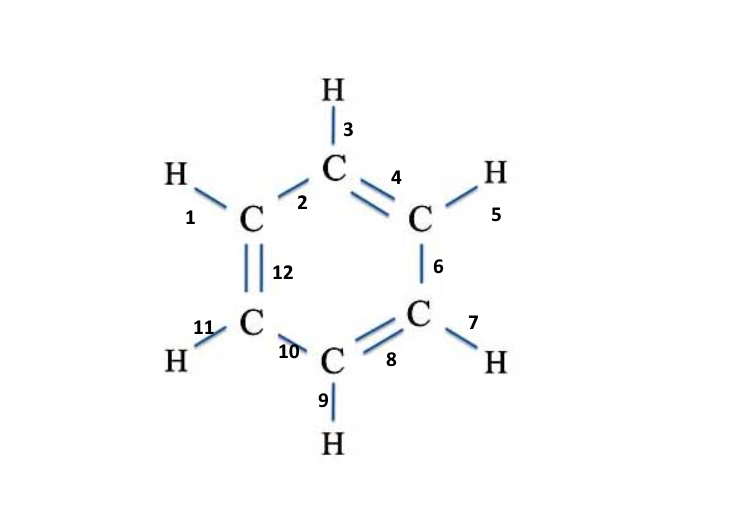
Other exceptions to the octet rule include boron-bearing compounds, which have a target of six shared electrons, rather than eight, because achieving an octet requires a formal charge. However, the double bond arrangement is preferred, as it reduces the overall formal charge on the atom. For some larger elements, the octet may be expanded to share more than eight electrons to reach a configuration with fewer formal charges.įor example, the sulfur atom in sulfur trioxide can form both single and double bonds with the surrounding oxygen atoms. Stable configurations generally minimize formal charges. Thus, the overall charge on the ammonium ion is one-plus. Since it is missing one electron, it bears a formal charge of one-plus. This leaves the nitrogen atom with four valence electrons instead of its usual five valence electrons. The formal charge is equal to the number of valence electrons on the neutral atom minus the number of bonds and unshared electrons on that atom.įor example, in the ammonium ion, each hydrogen atom has one valence electron in its neutral form and is bonded by a single bond, yielding a formal charge of zero. In such cases, a nonzero formal charge, F, is associated with the anomalous atom. Sometimes, atoms in polyatomic structures do not exhibit the standard number of valence electrons. With only twelve electrons available, they must all be bonding electrons to allow all atoms to reach a stable electronic configuration. Thus, twelve electrons must be shared between the atoms. It takes twenty-four electrons to satisfy the octets for two carbon atoms and the duets for four hydrogen atoms. Hence, ethene has a total of twelve valence electrons. Consider ethene, where two carbon atoms with four valence electrons each are surrounded by four hydrogen atoms having one valence electron each. Lewis structures are simplified representations of chemical bonds between atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
